Letter to German Health Minister, 13 December 2021

Letter to German Health Minister, 13 December 2021
13 December 2021
Prof. Dr. Karl Lauterbach MdB, Bundesgesundheitsminister
Deutscher Bundestag
Platz der Republik 1
11011 Berlin
Germany
Re: OFFICIAL COMPLAINT: ERN-Cranio; EU Clinical Practice Guidelines for Pierre Robin Sequence
Dear Prof. Dr. Karl Lauterbach, Federal Minister of Health,
We are writing to you with an official complaint concerning ERN-Cranio, which is managed by a Dutch healthcare provider, Erasmus Rotterdam Medical Center, and the EU Clinical Practice Guidelines which ERN-Cranio is allegedly producing for the rare disease, Pierre Robin Sequence. For the sake of EU babies born with this complex and life-threatening rare disease, we urgently request your immediate intervention in this time sensitive matter. We ask that you immediately contact the Dutch Minister of Health, Hugo de Jonge, and call upon the Dutch Minister (1) to immediately suspend all activities on this EU-funded project, which has been grossly mismanaged by Erasmus Rotterdam; and (2) start the project once again, this time including Germany’s top Pierre Robin Sequence Center of Expertise at the Tübingen University Hospital, and also including German patients, all of whom were unjustifiably excluded, in violation of EU rules.
Prof. Dr. Karl Lauterbach, Federal Minister of Health, as this is a matter of ongoing urgency, we would welcome the opportunity to meet with you or your advisors in Berlin or elsewhere to discuss this complaint in person. We look forward to hearing from you. Thank you.
Introduction
The EU’s European Reference Network (ERN) system was designed to represent the best of the best of Europe’s rare disease experts, across the entire EU. This year one of the ERNs, ERN-Cranio, launched a project to create official EU Clinical Practice Guidelines for the treatment of babies suffering from a life-threatening rare disease called Pierre Robin Sequence. These EU Clinical Practice Guidelines will be the first international medical guidelines ever produced for this complex rare disease. They will therefore have a significant impact on Pierre Robin Sequence babies for many years to come, and on the medical care these babies receive.
We, Pierre Robin Europe, are an official rare disease patient advocacy organization, an ePAG representative, in ERN-Cranio. We have served in this capacity since 2018. Our duty in ERN-Cranio is to promote the interests of babies suffering from the life-threatening rare disease, Pierre Robin Sequence. We have watched with increasing concern as this EU-funded project has been managed in a way which we believe violates EU rules, scientific rigor – a view shared by two leading international experts on this rare disease – and once again in an opinion held strongly by respected outside observers, common sense – based on ERN-Cranio’s arbitrary decision to exclude from the drafting group Europe’s two most respected Orphanet Centers of Expertise for this rare disease. Of the two Orphanet Pierre Robin Sequence Centers of Expertise which have been excluded, one is the Center of Expertise located in der Universitätsklinikum Tübingen, the Tübingen University Hospital.
ERN-Cranio: excluding the EU’s top experts in this rare disease
Prof. Dr. Christian Poets, Head of the Department of Neonatology at the Tübingen University Hospital, can confirm that ERN-Cranio has excluded Tübingen’s world class Pierre Robin Sequence Center of Expertise from this EU Guideline drafting project. Almost one year ago, Prof. Dr. Poets approached Dr. Irene Mathijssen, the Coordinator of ERN-Cranio at Erasmus Rotterdam Medical Center, spoke with her by telephone, and expressed Tubingen’s strong interest in joining the Guideline Development Group. Dr. Mathijssen would not permit Tübingen to join.
Dr. Mathijssen’s pretext for excluding the Tübingen University Hospital was that the EU Methodology which governs the creation of ERN Guidelines only permits members of an ERN to be included in an ERN’s Guideline Development Group. In fact, the EU Methodology contains no such rule. The EU Methodology states, in Handbook 4, Section 3.1, that the Guideline Development Group can include healthcare professionals who are members of the ERN, as well as healthcare professionals who are not members of the ERN.
Dr. Holm Graessner, the Coordinator of the European Reference Network for Rare Neurological Diseases (ERN-RND), Managing Director of the Center for Rare Diseases (ZSE) Tübingen, and Head of the research management unit of the Institute of Medical Genetics and Applied Genomics at the Tübingen University Hospital, can confirm that this rule in the EU Methodology which ERN-Cranio has invoked – which supposedly prohibits healthcare professionals who are not in an ERN from joining an ERN’s Guideline Development Group – does not in fact exist. The ERN system was built because rare disease knowledge is scarce; thus there exists an urgent need for European countries to pool resources and knowledge about each rare disease. It would make no sense whatsoever for the EU Methodology to prohibit world-class Orphanet Centers of Expertise in the EU from joining a Guideline Development Group, simply because they were not members of the ERN. And indeed, the EU Methodology contains no such rule.
Not only is Erasmus Rotterdam’s ERN-Cranio Coordinator Dr. Irene Mathijssen invoking a rule in the EU Methodology, when no such rule exists, but while Dr. Mathijssen is invoking this non-existent rule to exclude Tübingen, she has, at precisely the same moment, included in the Guideline Development Group the Great Ormond Street Hospital – and the Great Ormond Street Hospital is not a member of the ERN. As confirmed in writing by Dr. Mathijssen, the Great Ormond Street Hospital, which is not a member of ERN-Cranio, has played a leading role in this EU Guideline Development Project. How can the EU Methodology’s non-existent rule, which supposedly bars non-ERN healthcare professionals from participating in a Guideline Development Group, apply to Tübingen… but not apply to the Great Ormond Street Hospital? What we do know for certain is that Dr. Mathijssen’s hospital, Erasmus Rotterdam, has a relationship with the Great Ormond Street Hospital – they are friendly with one another. It is difficult to avoid the impression that Dr. Mathijssen and Erasmus Rotterdam have been running this EU-funded ERN Guideline development project based on relationships and politics, rather than on experience and expertise.
Making matters worse, Dr. Mathijssen and Erasmus Rotterdam, rather than honestly and openly admitting to errors in judgment, have instead engaged in an ongoing pattern of obfuscation and outright misrepresentation – not only with us, but also with their own government, the Dutch Ministry of Health. To take one example: on 15 September 2021 we received a letter from the Dutch Ministry of Health, which had been in contact with ERN-Cranio concerning these EU Guidelines. The Dutch Ministry of Health told us in this 15 September letter that according to ERN-Cranio, the Tübingen University Hospital was indeed involved in this EU Guideline development project. Not only was Tübingen involved, but Tübingen had even provided ERN-Cranio with feedback on a draft.
In reality, Tübingen (1) was absolutely not involved; and (2) the feedback which Tübingen tried to give to Erasmus Rotterdam was outright rejected, by Erasmus Rotterdam. On 8 September 2021, two days after the Tübingen University Hospital provided Erasmus Rotterdam with 17 pages of meticulously detailed scientific feedback on ERN-Cranio’s draft EU Guidelines for Pierre Robin Sequence – which had been written without Tübingen – Dr. Eppo Wolvius, of Erasmus Rotterdam, wrote the following message:
“Unexpectedly, I received a feedback form with comments on the RS Guideline from Prof. Poets, Tubingen. I am very surprised to receive these comments from a clinician outside the Steering Group of this Guideline initiated by the ERN CRANIO. Prof Poets informed me the draft was sent to him by Philippe Pakter. Maybe Philippe Pakter received the draft from you and therefore, I am sending you this mail. Please note that at this stage we are not allowed to involve centers outside the ERN CRANIO network.”
Erasmus Rotterdam’s claim that “we are not allowed to involve centers outside the ERN CRANIO network” was false on multiple levels. First of all, the EU Methodology contains no such rule. Second of all, Dr. Eppo Wolvius of Erasmus Rotterdam knew very well when writing this statement that the Great Ormond Street Hospital was involved in this ERN CRANIO project, because Dr. Wolvius was working on the Guidelines alongside the Great Ormond Street Hospital physicians – and yet the Great Ormond Street Hospital is “outside the ERN CRANIO network.”
Concerning the feedback itself, which Erasmus Rotterdam rejected: in the 17 pages of feedback the Tübingen University Hospital identified serious flaws, medical inaccuracies, misinterpreted research, unsupported conclusions, overlooked medical studies, and multiple forms of bias, in ERN-Cranio’s draft Guidelines.
Concerning bias in particular: in the feedback which Tübingen tried, unsuccessfully, to submit to Erasmus Rotterdam, Tübingen specifically protested the fact that ERN-Cranio’s draft Guidelines reflect a bias in favor of surgical interventions. Indeed ERN-Cranio actually states in their draft Guidelines that “The guideline steering group was chaired by a maxillofacial surgeon” and that “The guideline steering group consisted of eight professionals specialised in maxillofacial surgery and plastic surgery.” A bias toward surgery was inevitable.
This bias toward surgery constitutes a particularly grave defect in ERN-Cranio’s work, because while not all physicians agree on treatment strategies, virtually everyone agrees that surgery on newborn babies should only be performed when clinically necessary. The safe and medically proven Tübingen Palatal Plate treatment presents in the majority of cases a completely viable non-surgical alternative, which safely resolves the life-threatening upper airway obstruction associated with this rare disease. Nevertheless, Erasmus Rotterdam not only excluded the Tübingen University Hospital from the Guideline Development Group, but ERN-Cranio’s Coordinator Dr. Mathijssen, a surgeon, and Dr. Wolvius, a surgeon, would not even accept Tübingen’s feedback. It is worth adding here that while the draft Guidelines prepared by ERN-Cranio place an emphasis on surgical interventions, the clinical evidence demonstrating the efficacy and safety of the non-surgical Tübingen Palatal Plate treatment is actually stronger than the evidence in favor of surgery. Indeed the only Pierre Robin Sequence treatment which has ever been subjected to a controlled trial is the breakthrough non-surgical Tübingen Palatal Plate treatment developed by the Tübingen University Hospital.
Another form of bias which Tübingen protested in their feedback, which Erasmus Rotterdam refused to accept, concerned the following. According to ERN-Cranio’s draft, “Experts on each topic of the guideline were consulted to review the chapters and write recommendations.” What follows is a list of the 10 professionals who wrote the recommendations for these Guidelines. Every single one of the 10 professionals who wrote the all-important recommendations for these “EU” Guidelines come from the exact same country: the Netherlands. These are supposed to be Guidelines for the EU, not Guidelines for Holland. As Tübingen pointed out in their feedback, “It will automatically result in a strong bias if all experts invited are from only one country!”
And yet another form of bias which the Tübingen University Hospital specifically protested in their 17 pages of feedback – which Erasmus Rotterdam refused to accept – concerned the astonishing fact that as part of this EU-funded project’s patient survey for these EU Guidelines, patients from the EU’s largest Member State, Germany, were completely left out. This will be discussed in further detail below.
Most rare diseases lack an approved treatment or cure. The Tübingen Palatal Plate represents an exception, a shining hope: a safe, highly effective, economical rare disease treatment, developed right here in the EU. Stanford University has already adopted the Tübingen Palatal Plate treatment. Below is a photo of Dr. HyeRan Choo, Clinical Assistant Professor of Plastic and Reconstructive Surgery at Stanford University School of Medicine. Dr. Choo is holding a Tübingen Palatal Plate, which she and her colleagues learned how to produce and administer thanks to training from the Tübingen University Hospital:
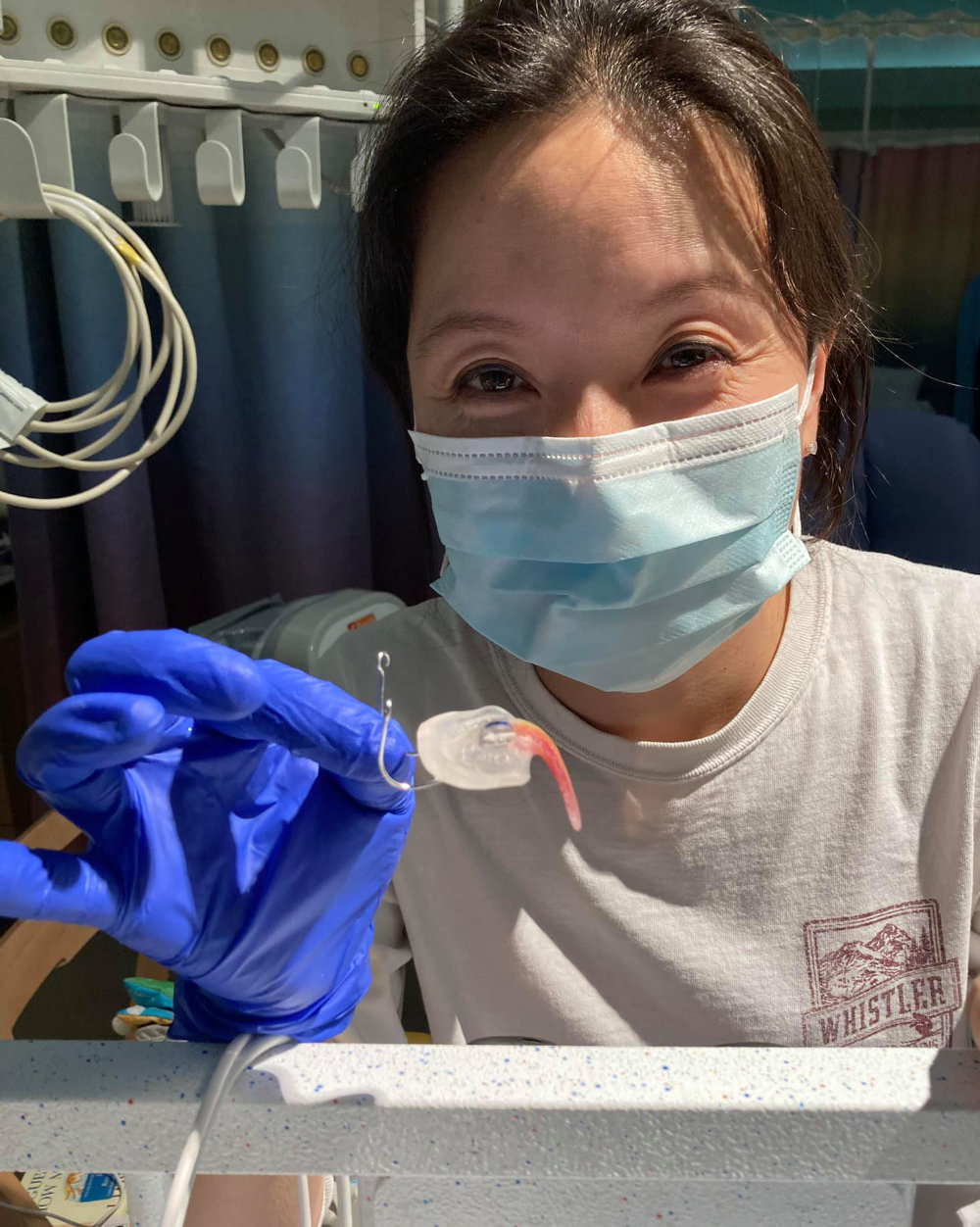
Dr. HyeRan Choo with a Tübingen Palatal Plate at Stanford
The American baby who received this particular Tübingen Palatal Plate at Stanford was able to safely avoid painful and risky surgery – labioglossopexy, and mandibular distraction osteogenesis. The mother wrote that “I truly believe this intervention needs to be shared with everyone with PRS.” With genuine excitement and hope, the mother described the treatment as “an incredible noninvasive option with great results. My daughter has only used the device 5 days and you can already see a vast improvement. Please reach out if you would like more information!!”
Soon, Harvard too will adopt the Tübingen Palatal Plate. This reflects well on the EU in general, and on German medicine in particular. Nobody in the international community of Pierre Robin Sequence experts questions the fact that the Tübingen University Hospital is one of the world’s foremost leaders in Pierre Robin Sequence treatment and research. And yet Erasmus Rotterdam, which Orphanet does not recognize as a Pierre Robin Sequence Center of Expertise, refused to even consider what Tübingen had to say about the many serious shortcomings in ERN-Cranio’s work.
Another example of Erasmus Rotterdam’s pattern of obfuscation and misrepresentation is the following. We recently received another letter from the Dutch Ministry of Health concerning these Guidelines, dated 30 November 2021. In this 30 November letter, the Dutch Ministry of Health indicates that according to Erasmus Rotterdam, the Guideline Development Group includes the following healthcare providers:
“Helsinki University Hospital, Vall d’Hebron Barcelona, Santa Maria Hospital Lisboa, Karolinska Hospital, Vicenza San Bortolo Hospital, Uppsala University Hospital, 12 de Octubre University Hospital Madrid, San Gerardo Hospital Monza, Hôpital Universitaire Necker, UMC Utrecht and Erasmus MC Rotterdam.”
The one healthcare provider which is missing from this list is the Great Ormond Street Hospital. Why did the Great Ormond Street Hospital suddenly disappear, when Erasmus Rotterdam’s Coordinator, Dr. Mathijssen, repeatedly confirmed that the Great Ormond Street Hospital is playing a leading role in this EU-funded project? We believe that the Great Ormond Street Hospital suddenly disappeared because the Great Ormond Street Hospital’s involvement in this project makes it unquestionably clear that Dr. Mathijssen and Erasmus Rotterdam have been managing this EU-funded project in an arbitrary and discriminatory manner.
Relationships, professional competition and politics are a known aspect of healthcare, just as they are a part of every other professional sector. However when they take hold of an important and sensitive EU-funded project to the extent that we see here, where the EU’s top two Pierre Robin Sequence Centers of Expertise are unjustifiably excluded from drafting EU Guidelines of care for this particular rare disease, then something must be done.
ERN-Cranio: excluding patients from the Guideline Development Group
In 2020 I was invited to submit an article to the Elsevier medical journal, “Seminars in Fetal & Neonatal Medicine.” One of the physicians who reviewed a draft of this rare disease patient perspective article wrote the following:
“Thanks, Philippe – this is tremendous and it emphasises the need or should I say absolute imperative that we obtain and heed the patient / parent perspective in the approach to rare diseases. This certainly provides aspects that we do not read about in the textbooks.”
(Prof. Dr. Peter A. Mossey, Professor and Personal Chair of Craniofacial Development & Dentofacial Orthopaeds, the University of Dundee; Associate Dean for Research; Director of WHO Collaborating Centre for Craniofacial Anomalies and Technology Transfer)
This rare disease patient perspective article which I submitted to Seminars in Fetal & Neonatal Medicine passed peer review and was published on 17 September 2021. Elsevier’s CEO, Ms. Kumsal Bayazit, was so impressed with the quality of the article that she personally waived the $4,100 Gold Open Access fee which Elsevier normally charges for Open Access in this particular medical journal; she also emailed me the following message:
“I am so very grateful you took the time and made the effort to write the paper from a rare disease patient and parent perspective. As you state it is critical that the vantage points and experiences of patients and parents are included in the literature to improve care and treatment.
…
My very best wishes to Lysiane and if I can be of any further assistance, please feel free to reach out to me.
Best regards,
Kumsal
Kumsal Bayazit
Chief Executive Officer
Elsevier”
Research makes it unquestionably clear that meaningful patient involvement in guideline development improves the quality of the final product:
“It has been shown that involvement of patients in guideline development improves the quality of guidelines and associated patient education materials and is particularly important when considering preference-sensitive decisions, for which patient preferences take priority when considering the benefits and harms of treatment options… Further challenges to patient involvement include ‘tokenistic’ involvement rather than routine integration of the patient voice, lack of support to help patients understand medical terminology, resistance from some health professionals, and a lack of resources.”
(Providing a Framework for Meaningful Patient Involvement in Clinical Practice Guideline Development and Implementation. Björkqvist, Josefine et al., European Urology Focus, Volume 7, Issue 5, 947 – 950.)
Unfortunately, ERN-Cranio’s coordinator Dr. Irene Mathijssen, in addition to excluding the Tübingen University Hospital from the Guideline Development Group, also excluded patients from the Guideline Development Group. Once again: ERN-Cranio’s EU Guideline Development Group included exactly zero patients.
The EU Methodology which governs the creation of EU Clinical Practice Guidelines for the ERN system absolutely requires that patients be included in the Guideline Development Group. The EU Methodology specifically identifies patients as one of the “four key constituents” of the Guideline Development Group. From the official EU Methodology, Handbook 4, Section 3.1:

The EU Methodology which governs the creation of EU Clinical Practice Guidelines for the ERN system absolutely requires that patients be included in the Guideline Development Group.
On 21 July 2021 a conference call was held with Dr. Mathijssen, a representative from Pierre Robin Europe, a representative from EURORDIS, and ERN-Cranio’s two Project Managers, to discuss the fact that ERN-Cranio had included no patients whatsoever in the Guideline Development Group for this EU Guideline development project. Various pretexts were given for the failure to include one single patient in the Guideline Development Group. One of the pretexts was that the EU Methodology was not yet available when ERN-Cranio began the project. Here are the facts.
Concerning the EU Methodology, the European Commission DG SANTE indicated to Pierre Robin Europe, in writing, in a letter dated 1 October 2021 (Ref. Ares(2021)5983971), that “The documents were initially distributed to all ERN coordinators in February 2021 in electronic format.” Once again, all ERN coordinators in all 24 of the ERNs received the EU Methodology in February. In a separate message, ERN-Cranio Project Manager Renée de Ruiter informed Pierre Robin Europe, in writing, in an email dated 14 July 2021, that ERN-Cranio launched this Guideline development project in February 2021.
Furthermore, in the European Commission DG SANTE’s abovementioned letter (Ref. Ares(2021)5983971), DG SANTE indicated that the European Commission was providing ERN-Cranio with €50.000 of financial support to develop these EU Clinical Practice Guidelines for Pierre Robin Sequence. We believe that ERN-Cranio channeled some portion or all of these EU funds to a private Dutch subcontractor named Qualicura (Emmastraat 2A, 4811 AG Breda, The Netherlands), in exchange for Qualicura’s assistance. If ERN-Cranio Coordinator Dr. Mathijssen did not have the time to read Handbook 4 of the EU Methodology governing the creation of EU Clinical Practice Guidelines for the ERN system, then she could have simply instructed the local Dutch subcontractor, Qualicura, to read it. After all, ERN-Cranio’s delegated duty is to produce EU Guidelines for the EU’s ERN system – not local Dutch guidelines based on local Dutch standards.
To summarize: in February 2021, ERN-Cranio’s clinicians began working on these EU Guidelines. In September 2021 the work was done, the ERN-Cranio clinicians had completed the assignment and written the document – about us, without us. But now we, the patients, the ultimate stakeholders, were permitted to take a look. Now we were permitted to provide comments, and this opportunity to provide comments on this document which we had not been allowed to participate in or contribute to in any way, at all, would presumably make the document “patient-centered.” Indeed in this document which we were now permitted to comment on in September, the ERN-Cranio clinicians had included the following proud declaration: “Since the guideline will be developed for patients and parents of patients, the patient perspective will be of major importance in this guideline.” It would be difficult to conjure up a more offensive example of base hypocrisy.
We sent the following feedback to Erasmus Rotterdam’s Dr. Eppo Wolvius:
“Stichting Pierre Robin Europe, ERN-Cranio’s official ePAG patient organization for Pierre Robin Sequence patients since 2018, believes this EU-funded project to develop Clinical Practice Guidelines for Pierre Robin Sequence (1) should be restarted from the beginning, and (2) should be run in accordance with the EU’s official ERN Methodology located at https://ern-ithaca.eu/documentation/guidelines/#ern-guidelines-methodological-handbook and (3) should include in the Guideline Development Group the EU’s top Pierre Robin Sequence Centers of Expertise. Our Board has formally concluded that accepting anything less would constitute a profound betrayal of the interests of the rare disease patients we are mandated to represent. Our 30 June 2021 letter to the Board of Member States, attached, and our 7 September 2021 letter to the European Commission/DG SANTE, attached, explain the many reasons for our position.”
Nobody from ERN-Cranio’s Guideline Development Group responded to our feedback. Dr. Eppo Wolvius did not even acknowledge receipt. We did not exist in February 2021, when the clinicians began working on this document, and we still didn’t exist in September 2021, when we were finally permitted to see what the clinicians had written, and present our justifiable grievances.
On Tuesday evening, 23 November 2021, Dr. Mathijssen called me, the director of Pierre Robin Europe, by telephone. During this telephone call, Dr. Mathijssen explained to me that although ERN-Cranio had not included any patients in the Guideline Development Group, ERN-Cranio had sent out a survey to patients.
First of all, Pierre Robin Europe, ERN-Cranio’s official ePAG patient organization for Pierre Robin Sequence since 2018, was not given any opportunity whatsoever to help create this patient survey. We were not even given the chance to look at the survey, before the survey was sent out. Apparently it never occurred to Dr. Mathijssen that ERN-Cranio’s official ePAG patient organization for Pierre Robin Sequence might want to see the Pierre Robin Sequence patient survey which the clinicians were going to send out, to patients. However, it gets worse.
The patient survey, which Pierre Robin Europe was not permitted to produce, or comment on, or even see, was sent out to patients by the Pierre Robin Sequence Centers of Expertise which were included in the Guideline Development Group. Thus, the survey was distributed to a specific subset of Pierre Robin Sequence patients: Pierre Robin Sequence patients who were fortunate enough to be in contact with a Pierre Robin Sequence Center of Expertise. If a patient had not been in contact with a Pierre Robin Sequence Center of Expertise, then there would be no way for them to access this survey.
Many, perhaps the majority, of poor patient outcomes arise in situations where a patient with a rare disease is unable to receive treatment at a Center of Expertise for that rare disease or complex condition. Based on the method which ERN-Cranio employed to distribute this survey, few if any of these patients would be reached, and none of their experiences or perspectives would be recorded. This is ironic, because the patient survey which the ERN-Cranio clinicians created – without us, the patients – was coordinated around the obstacles which patients encountered in the organization of Pierre Robin Sequence care, the difficulties they faced concerning the treatment plan, and organizational problems.
If ERN-Cranio had included Pierre Robin Europe, ERN-Cranio’s official ePAG patient organization for Pierre Robin Sequence, or indeed any patients at all, in the Guideline Development Group, as required by the EU Methodology, then we would have pointed out this serious sampling flaw. We would have offered to distribute the survey within our own patient community, using our international Pierre Robin Sequence patient network, thereby reaching patients who never reached a Center of Expertise. This would have enabled ERN-Cranio clinicians to obtain precious information on the most serious difficulties and obstacles patients faced, and thoughts from patients themselves on how such challenges might be addressed and resolved. This did not happen. And yet, it once again gets even worse.
Germany and German patients erased from Europe
Dr. Mathijssen stated during the 23 November 2021 telephone call that “it is very important to include all these various opinions and various experiences that people have in different countries, which differ a lot.” Therefore ERN-Cranio translated the patient survey into 8 European languages. The patient survey was even translated into Finnish, which is spoken in an EU Member State with a relatively low population of 5.5 million, and therefore an exceedingly small population of Pierre Robin Sequence patients.
However, there was one major EU language which the patient survey was not translated into: ERN-Cranio’s “EU” patient survey was not translated into German.
Germany has, by a very wide margin, the highest population of any Member State in the entire European Union, approximately 83 million people. We can safely assume that Germany has a far higher Pierre Robin Sequence patient population than any other country in Europe. For obvious reasons it would have made more sense to translate this survey into German, rather than translating it into Finnish or Portuguese. However ERN-Cranio, in defiance of financial prudence and common sense, decided that translating the survey into German was unnecessary.
Furthermore Dr. Mathijssen confirmed during this 23 November 2021 telephone call that this patient survey was not distributed to one single patient in the entire country of Germany. Germany’s entire Pierre Robin Sequence patient population, the largest Pierre Robin Sequence patient population in all of Europe, was completely left out.
These are supposed to be EU Guidelines for the European Union as a whole. Based on limited EU budgets, EU surveys should always be translated, first and foremost, into the major EU languages. This includes German. Furthermore, the ERN system is financed by the EU, and Germany’s financial contribution to the EU is far greater than the financial contribution of any other country in Europe. Based on the most basic notions of fairness and good faith, Germany should not be excluded from such EU projects. However, what is more shocking than anything else is that ERN-Cranio’s Coordinator, Dr. Mathijssen, who herself admitted that the various experiences people have in different countries “differ a lot,” thought that it was perfectly acceptable to run this EU project without including one single German healthcare provider, or one single German patient. ERN-Cranio simply removed the entire country of Germany, and Germany’s entire Pierre Robin Sequence patient population, from the map.
During the 23 November 2021 telephone call with Dr. Mathijssen I pointed out that in Germany, Pierre Robin Sequence babies can receive a highly specialised treatment which is not offered anywhere else in the entire European Union – the safe, medically proven, non-surgical, and cost-effective Tübingen Palatal Plate treatment. The Tübingen Palatal Plate treatment has now been adopted by Stanford University, which underwent training by the Tübingen University Hospital. The Tübingen Palatal Plate will soon also be administered by Harvard/Boston Children’s Hospital, which is currently undergoing training by the Tübingen University Hospital. And yet not one single Pierre Robin Sequence patient in Germany who received this breakthrough German treatment had the opportunity to fill out ERN-Cranio’s “EU” patient survey, or even learn of the existence of this “EU” survey.
A survey is not a substitute for a patient, or for any living human being. If meaningful diversity and inclusion could be achieved by sending out a survey, then there would be very few women or persons of color in EU governments or corporate boards. Instead of inclusion, the men in such positions would simply send out surveys. However for ERN-Cranio, even sending out one survey, to one German patient, proved to be too much.
When I asked Dr. Mathijssen how ERN-Cranio’s patient survey could achieve adequate coverage of EU patient experience, when it completely omitted the most populous Member State in all of Europe, Dr. Mathijssen replied that ERN-Cranio’s Guideline Development Group achieved German coverage through their literature review, which included medical studies published by the Tübingen University Hospital. Thus, ERN-Cranio can ignore the EU Methodology, which clearly requires patients in the Guideline Development Group, by instead sending out a survey; and in fact, even if it doesn’t send out a survey – not even one survey was sent out to a single one of Germany’s 83 million citizens and residents – that too is perfectly OK, because the “patient experience” can simply be extrapolated from clinical research, which is produced not by patients but by clinicians. Thus, according to Dr. Mathijssen’s logic, the EU’s ERN system can produce patient centered EU Guidelines without involving any patients or any patient surveys at all.
Dr. Mathijssen insisted during our 23 November 2021 phone call that ERN-Cranio’s EU Clinical Practice Guidelines for Pierre Robin Sequence conforms to the EU Methodology, and that ERN-Cranio’s performance on this project represents the “gold standard” of the EU’s ERN system. These were Dr. Mathijssen’s words: the “gold standard.” It is impossible to reconcile Dr. Mathijssen’s vision of this situation with objective reality.
Since early September 2021, we in Pierre Robin Europe, ERN-Cranio’s official Pierre Robin Sequence patient organization since 2018, have insisted that this EU Guideline development project for Pierre Robin Sequence be restarted from the very beginning. This EU-funded initiative must be relaunched and carried out as it should be carried out, in accordance with the EU rules, the official EU ERN Methodology – which the EU paid for, and distributed, to every single ERN in the entire ERN system. As ERN-Ithaca has rightfully pointed out, the EU Methodology “is an important step forward of the ERN CPG programme and a key element and tool for the work of the ERNs in the production, adaptation, adoption and appraisal of the different guidelines and technical sub-products, to be used for now on by all the ERNs. The methodology will ensure the quality and standardisation of the CPG and CDST produced and used by the ERNs, the backbone of the process of the diagnosis and treatment of patients suffering of rare or low prevalence and complex diseases.” ERN-Cranio’s performance on these Guidelines, and Erasmus Rotterdam’s mismanagement of the project, represent an embarrassing low point for the EU’s ERN system. Who pays the price? Babies suffering from a life-threatening rare disease, who are being cheated out the top quality EU Clinical Practice Guidelines which they desperately need.
Attached to this letter please find:
1) 30 June 2021 letter we sent to the Dutch Health Minister
This 30 June 2021 letter contains the most important facts, including the exclusion from the drafting group of the EU’s top two Centers of Expertise for Pierre Robin Sequence.
2) 7 September 2021 letter we sent to the Dutch Health Minister
In our 7 September 2021 letter to the Dutch Health Minister we demanded that this EU-funded initiative be relaunched and carried out as it should be carried out, in accordance with the EU rules, the official EU ERN Methodology, which the EU paid for, and distributed, to every single ERN in the entire ERN system.
3) the Dutch Ministry of Health’s response to us, 15 September 2021
In this 15 September 2021 letter from the Dutch Ministry of Health, the Dutch Ministry attempts to avoid responsibility for this matter, in spite of the fact that Erasmus Rotterdam’s conduct falls within the Dutch Ministry’s jurisdiction. The 15 September 2021 letter from the Dutch Ministry also contains a series of distortions and outright misrepresentations.
4) 30 September 2021 letter we sent to the Dutch Ministry of Health
In our 30 September 2021 letter we identified a series of factual and legal inaccuracies in the letter we received from the Dutch Ministry of Health on 15 September 2021.
5) 23 November 2021 letter we sent to Dr. Irene Mathijssen, Coordinator, ERN-Cranio
In this 23 November 2021 letter we inform Dr. Mathijssen that we have requested official intervention on the EU level.
On the evening of 23 November 2021, Dr. Mathijssen called me by telephone. We spoke about this situation involving the EU Guidelines for a little under 45 minutes.
6) 24 November 2021 letter we sent to Dr. Irene Mathijssen, Coordinator, ERN-Cranio
In this 24 November 2021 letter we sent to Dr. Mathijssen, we document our 23 November 2021 telephone call. During the 23 November 2021 telephone call, Dr. Mathijssen informed us that the Tübingen University Hospital had now been admitted to ERN-Cranio. Dr. Mathijssen said that since the Tübingen University Hospital was now a member of ERN-Cranio, the three healthcare providers which were in charge of this EU Guideline development project: Erasmus Rotterdam, Utrecht University Medical Center, and the Great Ormond Street Hospital – which is not a member of ERN-Cranio – would duly consider the Tübingen University Hospital’s 17 pages of feedback. Speaking on behalf of Pierre Robin Europe, the official ERN-Cranio ePAG patient organization for Pierre Robin Sequence, I made it clear to Dr. Mathijssen that this was not sufficient. The Tübingen University Hospital should have been included in the Guideline Development Group from the very beginning. The Tübingen University Hospital should have been involved in the discussion of the scope of these Guidelines. Tübingen should have been involved in the formulation of the clinical questions. Tübingen should have been involved in the discussion of the inclusion and exclusion criteria for selecting the relevant evidence. Tübingen should have been involved in the literature search and in the appraisal of the quality of the evidence. Tübingen should have been involved in the interpretation of the evidence, the construction of the recommendations, and the grading of the strength of the recommendations. The Tübingen University Hospital, one of the leading Pierre Robin Sequence Centers of Expertise in the entire world, should have been involved in the drafting of these EU Guidelines for Pierre Robin Sequence from day one, and it is an embarrassing scandal for the ERN system that they were senselessly kept out. Dr. Mathijssen’s belated assurance that she and her colleagues will now duly consider Tübingen’s feedback on a document which was produced completely without them is meaningless. At the end of this 24 November 2021 letter we sent to Dr. Mathijssen, we memorialize a proposal we had presented during the 23 November telephone call, as a possible way of moving forward. We proposed a conference call which would include Prof. Dr. Christian Poets of the Tübingen University Hospital, and another leading Pierre Robin Sequence expert, Prof. Dr. Corstiaan Breugem, who has probably published more medical studies on Pierre Robin Sequence than any other Pierre Robin Sequence expert in the world. At the end of our 23 November telephone call, Dr. Mathijssen solemnly promised that she would return to us with a response to our proposal “really soon.” Today is 13 December 2021; almost three full weeks have passed since our telephone call. Dr. Mathijssen has remained absolutely silent.
Additional material – my article, published in Seminars in Fetal and Neonatal Medicine
A parent’s view on the care of their baby with Robin sequence. Pakter, Philippe, Seminars in Fetal and Neonatal Medicine, Volume 26, Issue 6, 101288.
Conclusion
While ERN-Cranio’s Coordinator Dr. Irene Mathijssen and Erasmus Rotterdam are ultimately responsible for this dishonourable situation, which reflects so poorly on the EU’s ERN system, the Dutch Health Minister’s official inaction has played a pivotal role. The Dutch Ministry of Health’s ongoing justifications, rationalizations and misrepresentations have permitted Dr. Mathijssen to disregard our concerns, and to continue to “build facts on the ground,” with every passing month. Thus, when the issues which we in Pierre Robin Europe first raised in April of 2021 are finally discussed, the discussion will be meaningless, because the Guidelines will already be written. Perhaps this is the precise outcome which the Dutch Ministry of Health, Dr. Mathijssen, and Erasmus Rotterdam are looking to achieve.
For the sake of EU babies born with this complex and life-threatening rare disease, we urgently request your immediate intervention in this time sensitive matter. We ask that you immediately contact the Dutch Minister of Health, Hugo de Jonge, and call upon the Dutch Minister (1) to immediately suspend all activities on this EU-funded project, which has been grossly mismanaged by Erasmus Rotterdam; and (2) start the project once again, this time including Germany’s top Pierre Robin Sequence Center of Expertise at the Tübingen University Hospital, and also including German patients, all of whom were unjustifiably excluded, in violation of EU rules.
Prof. Dr. Karl Lauterbach, Federal Minister of Health, as this is a matter of ongoing urgency, we would welcome the opportunity to meet with you or your advisors in Berlin or elsewhere to discuss this complaint in person. We look forward to hearing from you. Thank you.
Kind regards,
Philippe Pakter
Pierre Robin Europe, chairman, pakter@pierrerobineurope.com
Member, EURORDIS, The European Organisation for Rare Diseases
Member, VSOP, Vereniging Samenwerkende Ouder-en Patiëntenorganisaties
PhD candidate, law, the University of Geneva: “Access to healthcare in Europe: the effectiveness of EU legislation in the context of rare disease patients”










